KARUPELV VALLEY PROJECT
Cyclic Dynamics in a Simple Vertebrate Predator-Prey Community
Olivier Gilg¹,²* Ilkka Hanski¹, Benoît Sittler³
The collared lemming in the high-Arctic tundra in Greenland is preyed
upon by four species of predators that show marked differences in the
numbers of lemmings each consumes and in the dependence of their dynamics
on lemming density. A predatorprey model based on the field-estimated
predator responses robustly predicts 4-year periodicity in lemming dynamics,
in agreement with long-term empirical data. There is no indication in
the field that food or space limits lemming population growth, nor is
there need in the model to consider those factors. The cyclic dynamics
are driven by a 1-year delay in the numerical response of the stoat and
stabilized by strongly density-dependent predation by the arctic fox,
the snowy owl, and the long-tailed skua.
| ¹Department of Ecology and Systematics,
Division of Population Biology, Post Office Box 65, 00014 University
of Helsinki, Finland. |
|
³Institut für Landespfiege,
University of Freiburg, 79085 Freiburg, Germany. |
²CBGP, Campus de Baillarguet,
Equipe Biologie et Gestion des Pullulations (INRA-IRD),
CS 30016, 34988 Montferrier/Lez Cedex, France. |
|
*To whom correspondence should be addressed.
Email: olivier.gilg@libertysurf.fr
|
| The cyclic dynamics of boreal and arctic populations of small
rodents is one of the most intensively studied phenomena in
population ecology. Many (1- 4), although not all (5, 6), researchers
now agree that the most likely mechanism that maintains cyclic
dynamics in boreal vole populations is predation by specialist
mustelid predators. In contrast, interaction with food resources
is thought to drive thedynamics of at least some lemming populations
(7). Even in the case of vole dynamics, competition among prey
for space or food is thought to play a key role in halting prey
population growth at high density, thereby allowing the predator
population to catch up with their faster-reproducing prey (1,
3, 8-10). One of the simplest vertebrate predator-prey communities
is that of lemmings and their dependent predators in the high-Arctic
tundra in Greenland. It constitutes only one mammalian prey,
the collared lemming (Dicrostonyx groenlandicus), and four predators,
the stoat (Mustela erminea), the arctic fox (Alopex lagopus),
the snowy owl (Nyctea scandiaca), and the longtailed skua (Stercorarius
longicaudus) (11, 12). The open tundra landscape and the continuous
daylight in summer in the high Arctic provide particularly favorable
conditions for fieldwork on vertebrate predators. We studied
the densities, breeding success, and diet of the four predators
in a 75-km² area in the Karup Valley in northeast Greenland
(72°30' N, 24°00' W), from 1988 to 2002. |
|
|
 |
| |
| Lemming |
|
| Lemming densities were estimated with live trapping for 1998
to 2002 (11, 13) and with regression between live-trapping results
and lemming winter nest counts (12, 14) for the other years
(15). The winter nests of lemmings are made of grass within
snow beds and are easily located on the ground after snowmelt.
We made a complete count of the nest numbers in an area of 15
km² every spring for 1988 to 2002. Although a varying number
of lemmings may use the same nest (16, 17), the winter nest
count in our large study area should closely reflect the actual
number of lemmings. The correlation between the spring densities,
as estimated by live trapping, and the winter nest count for
the years 1998 to 2002 is high (R² = 0.99, P < 0.01)
(fig. S1).
The stoat density was estimated from the number of lemming
winter nests predated and occupied by stoats in the 15-km²
area (12, 13). Stoats always use lemming nests in winter,
and stoat-occupied nests are easily distinguished by the abundance
of lemming fur
within the nest (12, 18).
Daily predation rates were plotted against the current (daily)
lemming density (N) to estimate functional responses of predators.
Daily predation rates were estimated from scat samples for
arctic foxes (n = 927) and stoats (n = 663), from direct observations
for
kuas (n = 475 hours), and from pellet samples and direct observations
for snowy owls
(n = 3419 pellets and 245 hours of observation). In the open
landscape and in the continuous daylight of summer, the behavior
of individual predators can be closely monitored over areas
as large as 5 km².
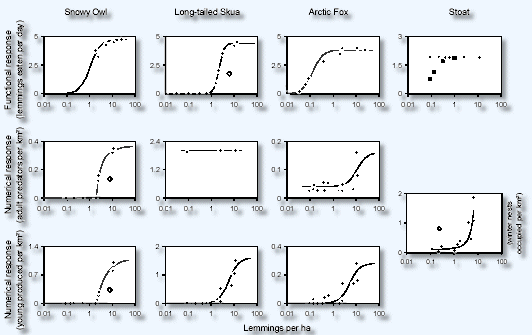
Predator densities were plotted against lemming density at
snowmelt to estimate numerical responses. With the exception
of the stoat, separate responses were estimated for adults
and weaned or fledged young (15). The daily consumption rate
of the avian predators is somewhat higher than that of the
mammalian predators, but the latter are more efficient at
catching prey at low lemming densities ( predation half-saturation
constant < 0.2 lemmings/ha) than are the former (≥1)
(Fig. 1, upper row). The numerical responses of the predators
are species specific. The nomadic snowy owl only settles and
breeds in areas where lemming density at snowmelt (N') exceeds
a threshold of ≈ 2 lemmings/ha. The constant adult density
in summer of the migratory long-tailed skua is five times
as high as that of the snowy owl when the latter is present,
but the skua breeds successfully only when N > 1. The arctic
fox shows elevated breeding success when N > 1 but maintains
a relatively constant adult density, except in peak lemming
years (N ≈10) (Fig. 1, middle row), when fox density
may increase greatly.
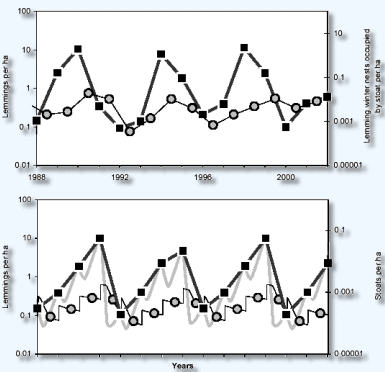
The stoat is the only predator that shows a delayed response
to changes in prey density, with highest numbers seen the
year after the lemming peak (Fig. 2, top). The results for
the stoat in Fig. 2 are based on the counts of lemming winter
nests occupied by the stoat, rather than on population size
estimates, but our field observations support the assumption
that the winter nest count gives a reliable picture of the
long-term dynamics (15).
The results in Fig. 2 demonstrate a 4-year cycle in lemming
and stoat numbers from 1988 to 1998. The years 1999 to 2002
show a somewhat deviating pattern, possibly indicating that
this cycle will be 5 years long. The occurrence of 4-year
population cycles in lemmings is supported supported by another
source of information. The trapping records for the arctic
fox in northeast Greenland from 1935 until 1960 showed distinct
peaks in the winters of 1937-38, 1941-42, 1946-47, 1950-51,
1954-55, and 1958-59 (19), at intervals of 4, 5, 4, 4, and
4 years. These trapping records most likely reflect changes
in lemming density. What is maintaining the regular 4-year
lemming cycle? We have constructed a model to elucidate the
role of predation in lemming dynamics. In our predator-prey
model, the functional responses of the snowy owl, the longtailed
skua, and the arctic fox are dependent on N, and their numerical
responses are determined by N'. Hence, these responses are
strict functions of spring and current (daily) lemming densities,
and the impact of these predators on the lemming population
is implemented in the model through the functions in Fig.
1 (15). The responses of these predators are effectively density-
dependent constants and, once estimated from field data (table
S1), were never altered in our modeling.
The dynamic part of the model involves the lemming and the
stoat. Reproduction in the collared lemming is continuous,
with overlapping generations and with a higher rate of reproduction
in winter than in summer (11, 20- 22). The maximum observed
lemming densities were ≈10 individuals per ha (Fig.
2, top). This density could potentially be much higher (11,
23-25). There is an excess of burrows available for lemmings
in our study area, and there is no evidence that food is a
limiting factor (15). Because there is no evidence for space
or food limitation, lemming dynamics were modeled by continuous-time
exponential growth, with different growth rates for winter
(r/w) and for summer (r/s, from June 15 to September 25).
The dynamics of the stoat were modeled by assuming that all
females produce one litter per year and that stoat mortality
is an S-shaped function of lemming density (15).
|
|
|
 |
| Lemming |
| |
| |
| |
| |
| |
| |
| |
 |
| Young foxes |
| |
| |
| |
| |
| |
| |
| |
 |
| Harfangs |
| |
| |
| |
| |
| |
| |
| |
 |
| Skua |
| |
| |
| |
| |
| |
| |
| |
 |
Mustela
Photos © GREA & Olivier Gilg |
|
|
Figure 1
Predators' responses to lemming density. Functional responses (top)
are related to the current (daily) lemming density (N), whereas the
numerical responses (middle and bottom) are related to the lemming
density at snowmelt (N ). The numerical response of the stoat includes
adults and weaned young and is delayed (x axis is the average lemming
density at snowmelt for the past 2 years). The functional response
of the stoat (top right) shows estimates based on winter (diamonds)
and summer (square) scats. Open symbols are outliers resulting from
unusual climatic or demographic situations (15). The numerical response
of adult skuas could only be assessed for 1998 to 2000, when we arrived
at the study area in the beginning of June and before the skuas started
to ?ock. The species is known to have stable densities (28).
Figure 2
 |
|
Empirical (top) and model-predicted (bottom) time series for
the lemming (squares) and the stoat (circles). Data points for
the lemming are at snowmelt and for the stoat at midwinter.
The lemming density estimate was calculated on the basis of
absolute lemming winter nest counts for 1988 to 2002 and live-trapping
density estimates for 1998 to 2002 (15). The gray line (bottom)
shows the lemming dynamics predicted by the model; the bold
black line represents the overall lemming dynamics to be compared
with the empirical time series. There is a correspondence in
cycle length, amplitude, and maximum lemming densities. Parameter
values for the predicted series: r/s 0.8, r/w = 4, v = 4, c
= 1000, D = 0.1, N/crit = 0.1, d/low = 0.1, d/high = 4, and
b = 25. v, number of weaned stoats produced per female per year;
c, maximum per capita predation rate of the stoat, in lemmings
per year; b, slope of the mortality function for the stoat (table
S2). |
Figure 3
Cycle lengths and maximum densities predicted by 81 parameter
combinations given in table S2. Two combinations are not included
that led to noncyclic dynamics or to the lemming population
increasing exponentially. Circle width is proportional to the
number of parameter combinations yielding the cycle length and
maximum lemming density indicated in the ?gure (smallest circle,
1 parameter combination; largest circle, 17 combinations). |
|
 |
|
|
R E P O R T S
Predation by stoat was modeled with a type III functional response
(3) to incorporate the possibility of stoats finding alternative
prey at lowest lemming densities, when lemmings are so dispersed
(less than 10 per km²) that they must become very hard for
stoats to locate (15). In this situation, with stoat density ≥
1 per 15 km², it would be unrealistic to use a type II functional
response, which assumes a steeply increasing predation rate for
the lowest prey densities.
We analyzed the model numerically by keeping the parameters for
the arctic fox, the snowy owl, and the long-tailed skua fixed at
the values estimated in Fig. 1 and reported in table S1. Of the
remaining parameters (table S2), the summer growth
rate of the lemming (r/s), the predation halfsaturation constant
of the stoat (D), and the mortality rate parameters of the stoat
(dlow, dhigh, and Ncrit) (15) have more uncertainty than the others.
We ran model simulations for many combinations of these parameters
(table S2).
The model mostly predicts complex dynamics, but with a strong regular
component, which most frequently has a period of 4 years (Figs.
2 and 3). The most notable difference between the predicted and
observed dynamics is in the shape of the cycle: The increase phase
of the cycle appears to be shorter in empirical results than in
model predictions (Fig. 2).
Turchin et al. (7) have argued that Norwegian lemming oscillations,
characterized by
sharp and angular peaks, are driven by interactions with food plants,
but our study suggests
that similarly sharp peaks can also be observed for predator-regulated
lemming populations. The notable feature of the present modeling
results is that there is no food or space limitation in the lemming
dynamics; in other words, there is no intrinsic prey density dependence.
This is supported empirically, because there is no evidence for
food or space limitation in the field, or for any other mechanism,
apart from predation, contributing to population regulation. The
predicted dynamics are generated by a combination of destabilizing
predation by the stoat and strongly stabilizing predation by the
three other species of predators. This is an example of cyclic predator-prey
dynamics in which the prey dynamics are entirely determined by predation.
If one of the avian predators is completely removed from the model,
the dynamics change: The lemming escapes from the control of the
predators. If removed, the arctic fox is less influential to lemming
dynamics, which remain qualitatively unchanged, unless the summer
growth rate of the lemming (r/s) is higher than 0.25 (which value
is within the feasible range for r/s) (table S2). Removing the stoat,
which has a much lower predation rate than the avian predators for
most of the cycle (Fig. 1), does not allow the lemming to escape
predator control as long as r/s < 0.33.
However, removing the stoat leads to noncyclic dynamics, because
the delayed numerical response of the stoat is the driving force
of the multiannual fluctuations. In this simple community, all the
predators have distinct life histories and responses to changing
prey population size, which should help them coexist on the single
prey (26, 27).
|
References and Notes
1. I. Hanski, H. Henttonen, E. Korpima¨ki, L. Oksanen, P.
Turchin, Ecology 82,1505 (2001).
2. T. Klemola, T. Pettersen, N. C. Stenseth, Advances in
Ecological Research 33, 75 (2003)
3. P. Turchin, Complex Population Dynamics (Princeton
Univ. Press, Princeton, NJ, 2003).
4. P. Turchin, I. Hanski, Ecology Letters 4, 267 (2001).
5. X. Lambin, C. J. Krebs, R. Moss, N. G. Yoccoz, in Population
Cycles: The Case for Trophic Interactions, A. A. Berryman, Ed.
(Oxford Univ. Press, Oxford, 2002), pp. 155-176.
6. M. K. Oli, Trends Ecol. Evol. 18, 105 (2003).
7. P. Turchin, L. Oksanen, P. Ekerholm, T. Oksanen, H.
Henttonen, Nature 405, 562 (2000).
8. I. Hanski, P. Turchin, E. Korpima¨ki, H. Henttonen,
Nature 364, 232 (1993).
9. I. Hanski, E. Korpima¨ki, Ecology 76, 840 (1995).
10. P. Turchin, I. Hanski, Am. Nat. 149, 842 (1997).
11. O. Gilg, Oikos 99, 499 (2002).
12. B. Sittler, Ann. Zool. Fennici 32, 79 (1995).
13. O. Gilg, thesis, Universite´ de Franche-Comte´
(2002).
14. B. Sittler, O. Gilg, T. B. Berg, Arctic 53, 53 (2000).
15. Materials and methods are available as supporting
material on Science Online.
16. F. B. Chernyavsky, in Materials on the Ecology of
Small Subarctic Mammals, F. B. Chernyavsky, Ed.
(Nauka, Novosibirsk, 1975), pp. 13-33.
17. T. N. Dunaeva, Proceedings of the Institute of Geography,
Academy of Sciences of the U.S.S.R. 51, 78 (1948).
18. B. M. Fitzgerald, J. Anim. Ecol. 46, 367 (1977).
19. P. Schmidt Mikkelsen, Nordøst-Grønland 1908-1960:
Fangstmanperioden (Dansk Polarcenter, Copenhagen,
1994). |
|
20. T. N. Dunaeva, Bulletin of the Moscow Society for Nature
Protection, Biological Department 83, 18 (1978).
21. F. B. Chernyavsky, S. P. Kiriushchenko, T. V. Kiriushchenko,
Ecology of Birds and Mammals from Wrangel Island: Material Concerning
the Winter Ecology of Siberian Lemming (Lemmus sibiricus) and
Collared Lemming (Dicrostonyx torquatus) (Nauka, Vladivostok,
1981).
22. J. S. Millar, Ecoscience 8, 145 (2001).
23. G. O. Batzli, R. G. White, S. F. MacLean, F. A. Pitelka,
B. D. Collier, in An Arctic Ecosystem: The Coastal
Tundra at Barrow, Alaska, J. Brown, P. C. Miller, L. L.
Tieszen, F. L. Bunnell, Eds. (Dowden, Hutchinson, &
Ross, Stroudsburg, PA, 1980), pp. 335-390.
24. T. B. G. Berg, thesis, University of Copenhagen (2003).
25. G. H. R. Henry, J. Svoboda, B. Freedman, Can. J. Bot.
68, 2660 (1990).
26. P. Chesson, Theor. Pop. Biology 45, 227 (1994).
27. P. Chesson, Ann. Rev. Ecol. Systematics 31, 343 (2000).
28. M. Andersson, J. Anim. Ecol. 45, 537 (1976).
29. We thank R. A. Ims, X. Lambin, P. Delattre, J. Sundell,
O. Ovaskainen, and anonymous referees for discussion and comments
on the manuscript and B. Sabard, A. Hurstel, R. Sane´,
and E. Buchel for help in the ?eld. This work was supported
by the French Polar Institute (IPEV ), Academy of Finland (the
Finnish Centre of Excellence Programme 2000-2005, grant 44887),
Deutsche Forschungsgemeinschaft, and Groupe de Recherches en
Ecologie Arctique. Dansk Polarcenter and Sirius sledge patrol
helped with the logistics. |
|
Extracted to "Science"
Supporting Online Material
www.sciencemag.org/cgi/content/full/302/5646/866/DC1
Materials and Methods
Fig. S1
Tables S1 and S2
2 June 2003; accepted 5 September 2003
|